COVID-19 vaccines: what you should know
COVID-19 vaccines have been topical and continue to raise several questions from the public, including when South Africa will receive vaccines, who will receive it first, and why vaccination is important.
28 January 2021
COVID-19 vaccines: what you should know
COVID-19 vaccines have been topical and continue to raise several questions from the public, including when South Africa will receive vaccines, who will receive it first, and why vaccination is important.
28 January 2021
To find out more about vaccination in general, COVID-19 vaccines and how South Africans will benefit, see the following frequently asked questions compiled by the National Institute For Communicable Diseases (NICD):
COVID-19 vaccines
1. Is there a vaccine for COVID-19?
Yes, there are multiple COVID-19 vaccines. Certain vaccines from some countries’ regulatory authorities have been approved for use and many more COVID-19 vaccines are currently being developed. Once vaccines are demonstrated to be safe and effective, the World Health Organization (WHO) collaborates with global partners to ensure equal access to COVID-19 vaccines for the billions of people who will need it. Most COVID-19 vaccines being tested or reviewed require two doses.
2. How are the COVID-19 vaccines being distributed around the world?
The first COVID-19 vaccines are currently being introduced in certain countries. Before COVID-19 vaccines can be delivered, the vaccines must be proven safe and effective in large clinical trials. For the WHO to consider a vaccine, a series of independent reviews relating to the efficacy and safety must be done, including regulatory review and approval in the country where the vaccine is manufactured.
In addition to reviewing the data for regulatory purposes, the evidence must also be reviewed for policy recommendations on how the vaccines should be used. An external panel of experts convened by WHO, the Strategic Advisory Group of Experts on Immunization (SAGE), analyses the results from clinical trials, along with evidence on the disease, age groups affected, risk factors for disease, and other information. The panel then recommends whether and how the vaccines should be used. Thereafter officials in individual countries decide whether to approve the vaccines for use in their country and if approved they develop policies for how to use the vaccines based on the WHO recommendations.
The vaccines must be manufactured in large quantities, an unprecedented challenge, as other important life-saving vaccines (already in use), also continue to be produced. As a final step, all approved vaccines will require distribution through a complex logistical process, with rigorous stock management and temperature control.
3. How quickly could COVID-19 vaccines stop the pandemic?
Several factors will determine the impact of vaccines on the COVID-19 pandemic. These factors include the effectiveness of the vaccines; how quickly they are approved, manufactured, and delivered; and how many individuals are vaccinated.
As is the case with all vaccines, most scientists anticipate that COVID-19 vaccines will not be 100% effective. The WHO will help ensure that any approved vaccines are as effective as they can be, to have the greatest possible impact on the pandemic.
4. Will COVID-19 vaccines provide long-term protection?
Presently it’s too early to know if COVID-19 vaccines will provide long-term disease protection, as additional research needs to be conducted. However, the data available suggests that most individuals who recover from COVID-19 develop an immune response that provides some period of protection against reinfection. How strong this protection is and how long it lasts, is not yet clear.
5. What is COVAX?
COVAX is a global alliance bringing together governments, global health organisations, manufacturers, scientists, the private sector, civil society and philanthropy, to provide innovative and equitable access to COVID-19 vaccines. This will ensure that individuals across the globe, regardless of financial or societal status, will have access to COVID-19 vaccines once available.
6. How will the vaccines be allocated to different countries?
Once the COVAX vaccines have proved to be safe, effective, have successfully undergone clinical trials, and have received regulatory approval, available doses will be allocated to participating countries at the same rate, proportional to their total population size.
No country will receive enough doses to vaccinate more than 20% of its population until all countries in the COVAX group have been offered this amount.
COVID-19 vaccines in South Africa
1. When will South Africa receive vaccines?
South Africa will receive one million doses of the COVID-19 vaccine in January and 500 000 doses in February of the Oxford University-AstraZeneca vaccine from the Serum Institute of India (SII).
2. What else is South Africa doing to get vaccines?
An agreement has been reached with the COVAX Facility to secure vaccines to immunise 10% of the population. These doses are expected at the beginning of the second quarter of the year and every effort is made to work with various pharmaceutical companies to ensure 67% of the population is vaccinated by the end 2021. The country’s membership in the COVAX Facility ensures that South Africa receives its equitable share of the vaccines as it becomes available.
3. Will these vaccines have to be approved before they can be used in South Africa?
Yes. Before a vaccine can be distributed, the South African Health Products Regulatory Authority (SAHPRA) must assess the safety, efficacy and quality of the vaccine. SAHPRA has committed to the expeditious evaluation of these vaccines through various mechanisms. The Oxford University-AstraZeneca vaccine has already been approved by various regulators around the world and is being distributed in other countries.
4. Does South Africa have a strategy to distribute the vaccines once we receive them?
Yes. The vaccine will be rolled out in a three-phased approach.
5. Who in South Africa will be vaccinated first?
The country’s estimated 1,25 million frontline healthcare workers will receive the vaccine first. Phase 2 will focus on essential workers, persons in congregate settings, persons over 60 years and persons over 18 years with co-morbidities. The final phase will target 22,5 million members of the population over the age of 18 years.
6. Who will manage the rollout of the vaccine strategy?
The Government will source, distribute and oversee the rollout of the vaccine. As the sole purchaser of vaccines, the Government will distribute it to provincial governments and the private sector.
A national register for COVID-19 vaccinations will be established. The vaccination system will be based on a pre-vaccination registration and appointment system. All those vaccinated will be placed on a national register and provided with a vaccination card. A national rollout committee will oversee the vaccine implementation in both the public and private sectors.
Source: https://www.nicd.ac.za/covid-19-vaccines-what-you-should know/?utm_content=buffered29b&utm_medium=social&utm_source=twitter.com&utm_campaign=buffer
How do COVID-19 vaccines compare?
There are several COVID-19 vaccines in various stages of authorisation, approval and development around the world.
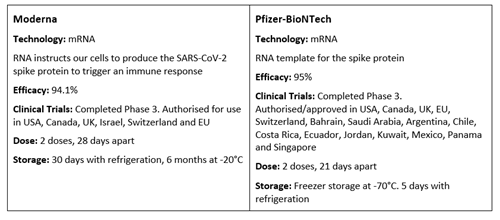
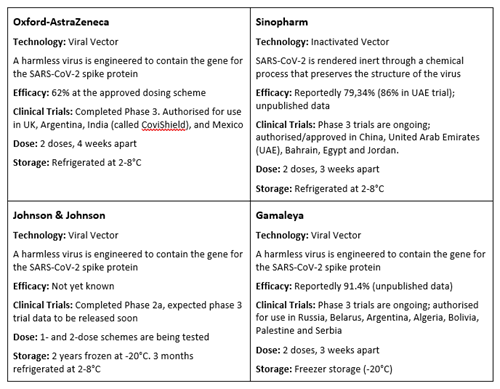
Source: The Unbiased Science Podcast | @unbiasedscipod
Important contact numbers
Fedhealth Customer Contact Centre: Monday to Thursday 08h30 – 17h00; Friday 09h00 – 17h00.
Call 0860 002 153 or email member@fedhealth.co.za
Fedhealth Nurse Line and Emergency Medical Services: 0860 333 432 Paed-IQ: 0860 444 128 and SOS Call Me: *130*3272*31# Press 1. Emergency Medical Services; 2. Nurse Line; 3. MediTaxi
Visit the Coronavirus Hub on the Fedhealth website: https://fedhealth.co.za/media/2338/fedhealth-coronavirus-faqs-document.pdf or use the COVID-19 Chatbot (go to LiveChat on fedhealth.co.za to select). You can also use the WhatsApp service (click on https://wa.me/27600702479 from your mobile device) for assistance.
Important COVID-19 resources
COVID-19 online resource and news portal: https://sacoronavirus.co.za/
The National Institute for Communicable Diseases (NICD): https://www.nicd.ac.za/